Veridica — Clinical Data Intelligence Platform
Overview
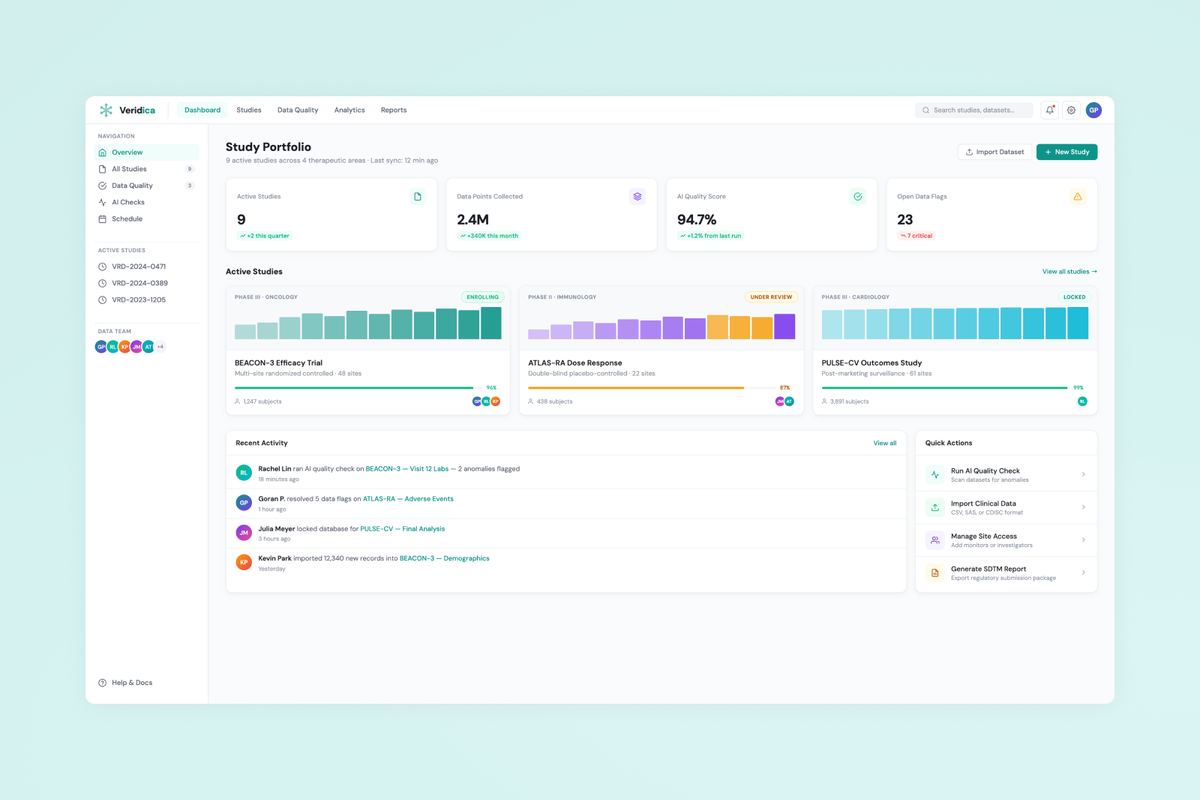
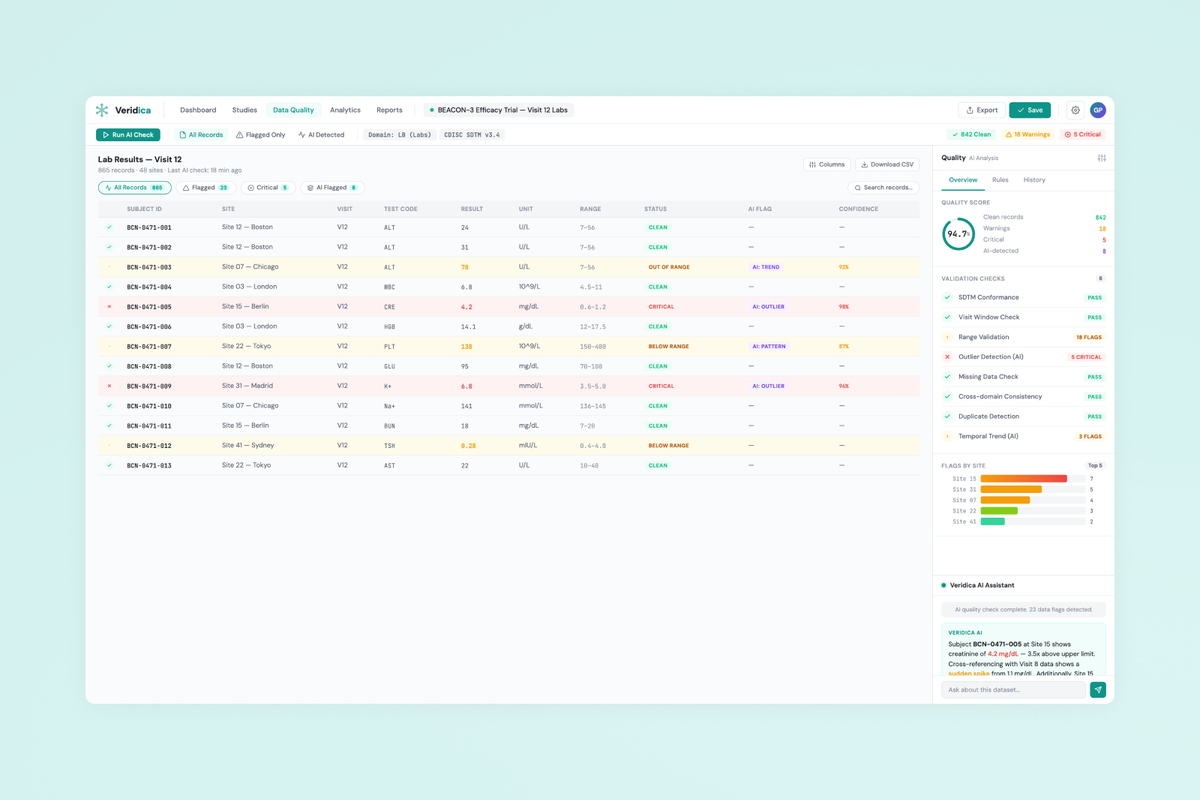
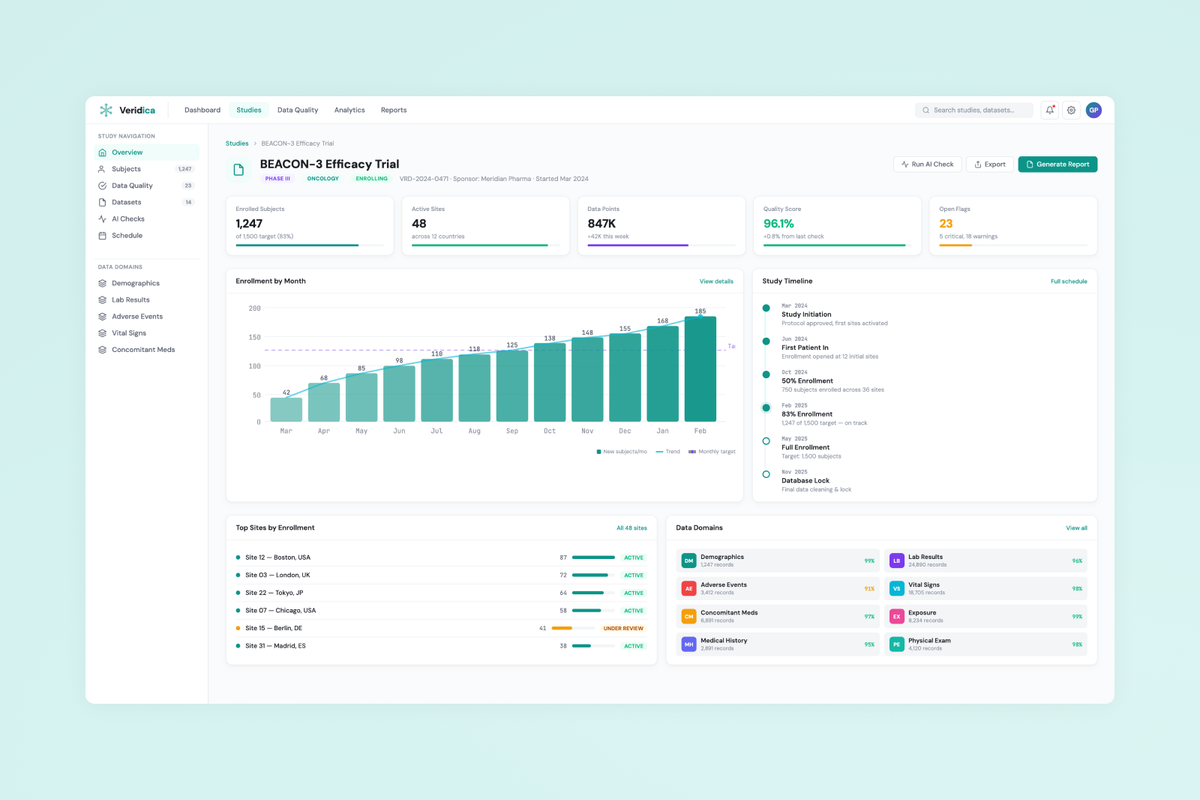
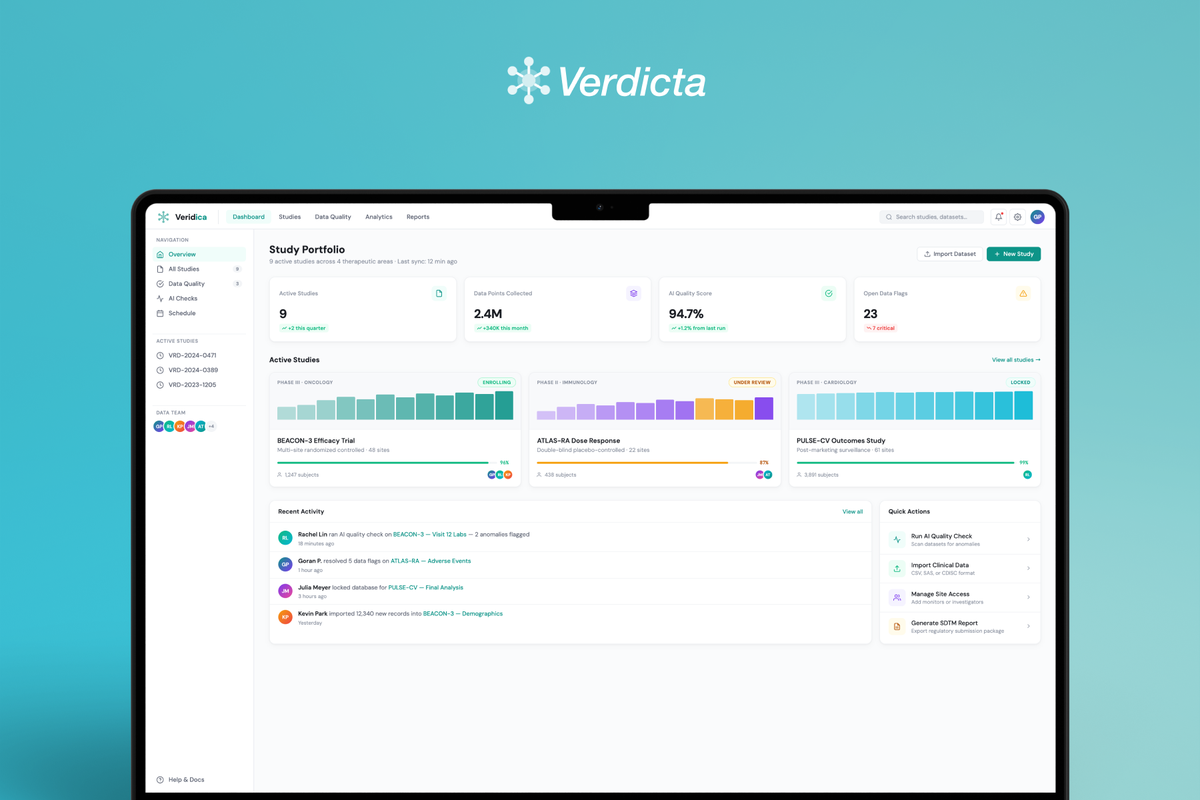
Veridica is a clinical trial data management platform that aggregates data from multi-site studies and uses AI to detect quality issues, anomalies, and compliance gaps — replacing fragmented spreadsheet workflows with a single intelligent command center for clinical operations teams.
Project brief
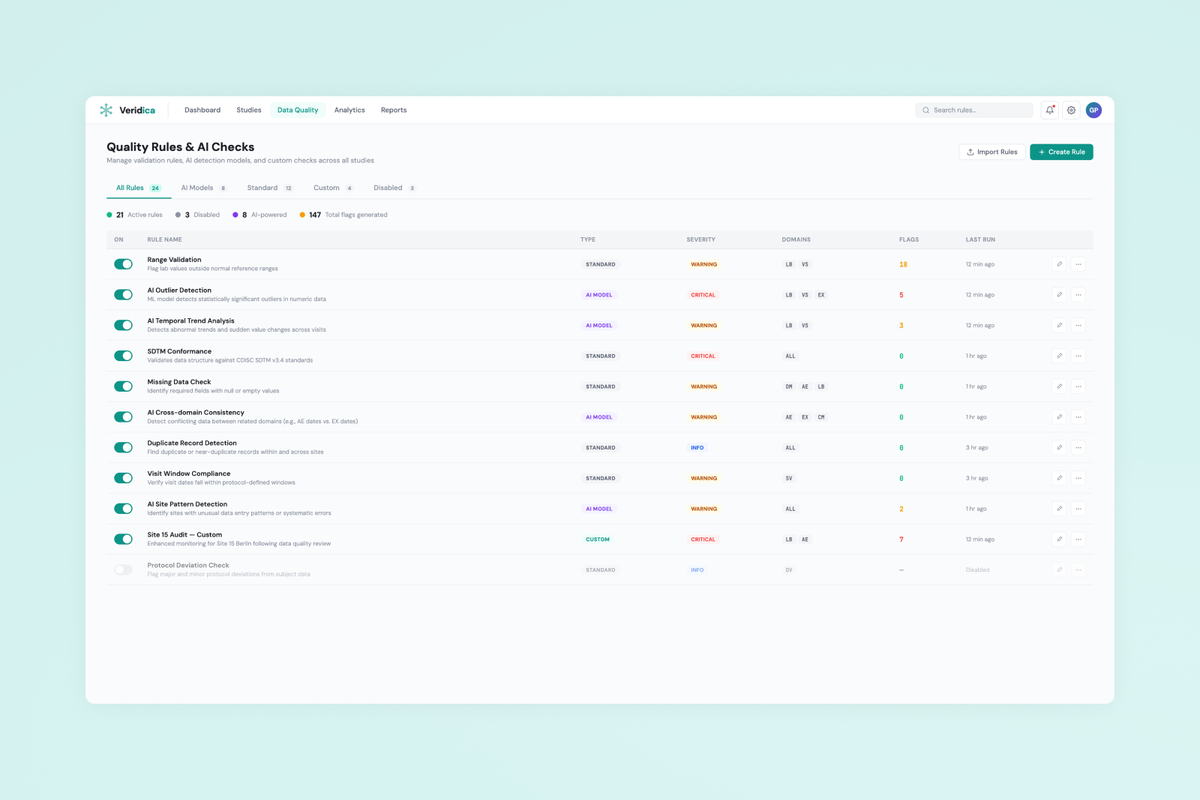
Design a B2B SaaS platform that gives clinical data managers real-time visibility into trial data quality across sites, with AI-powered anomaly detection and configurable compliance rules aligned to CDISC/SDTM standards.
Role
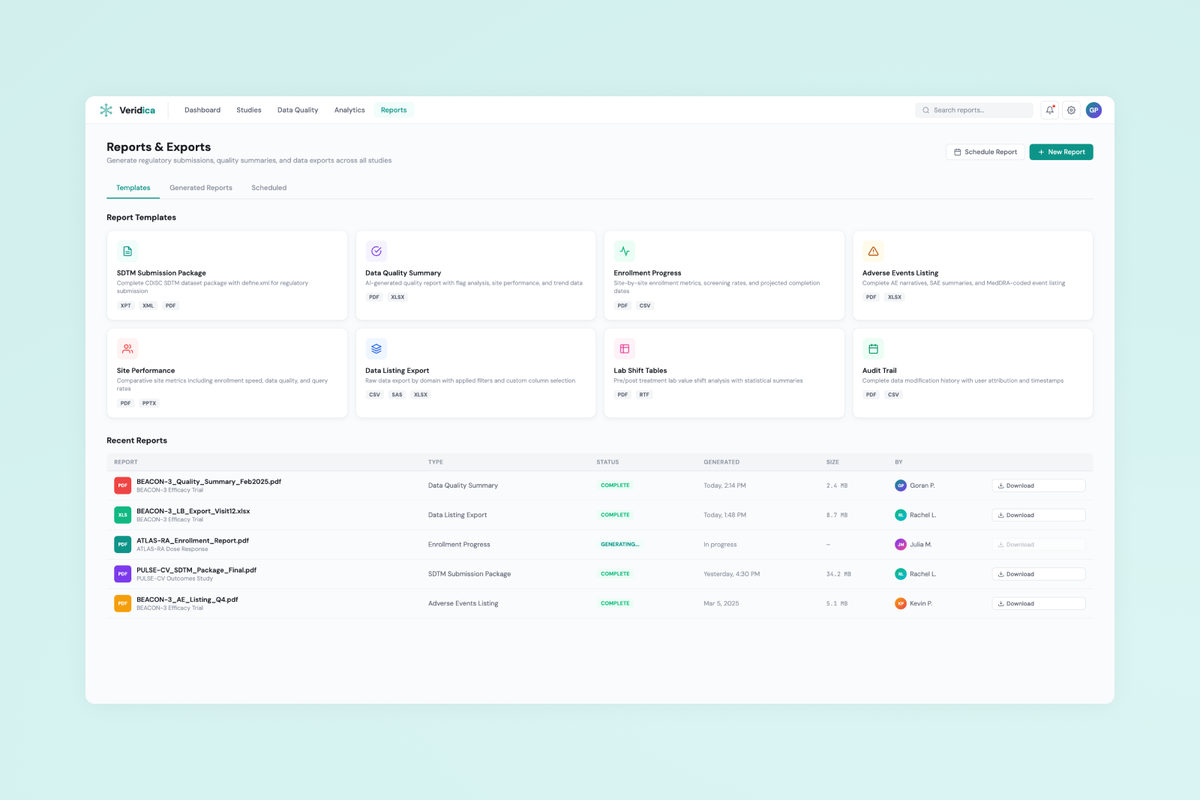
Led the full design cycle over 8 months — from research and information architecture through a Figma design system and production handoff with a React/TypeScript engineering team. Key deliverables: Dashboard, Data Quality Review, Study Overview, AI Rules Configuration, and Reporting.
Methods
- Discovery interviews with clinical data managers and biostatisticians to map pain points in multi-site data reconciliation.
- Information architecture for a dashboard-first experience surfacing anomalies, compliance gaps, and site-level trends.
- Data visualization design for complex clinical datasets — balancing density with clarity across tables, charts, and rule-based views.
- AI rules configuration UX allowing non-technical users to create, tune, and audit automated quality checks.
- Design system built in Figma and handed off as tokenized specs to a React/TypeScript engineering team.
Work